 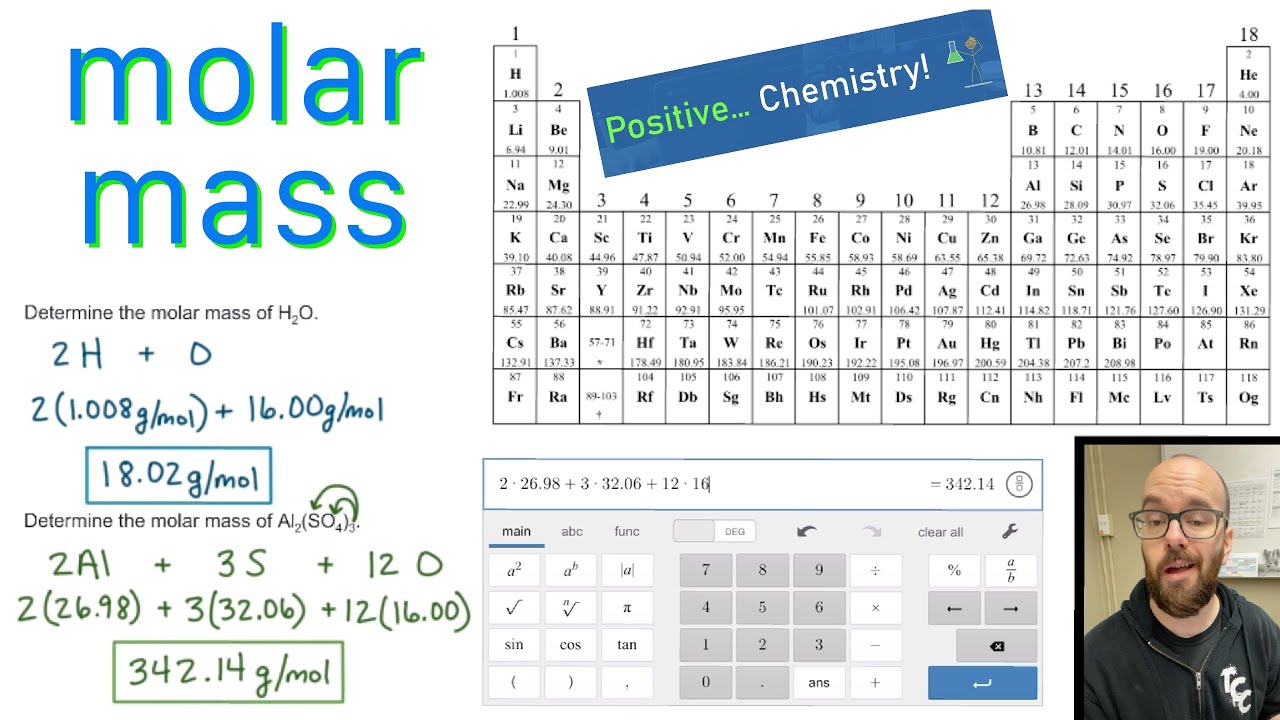 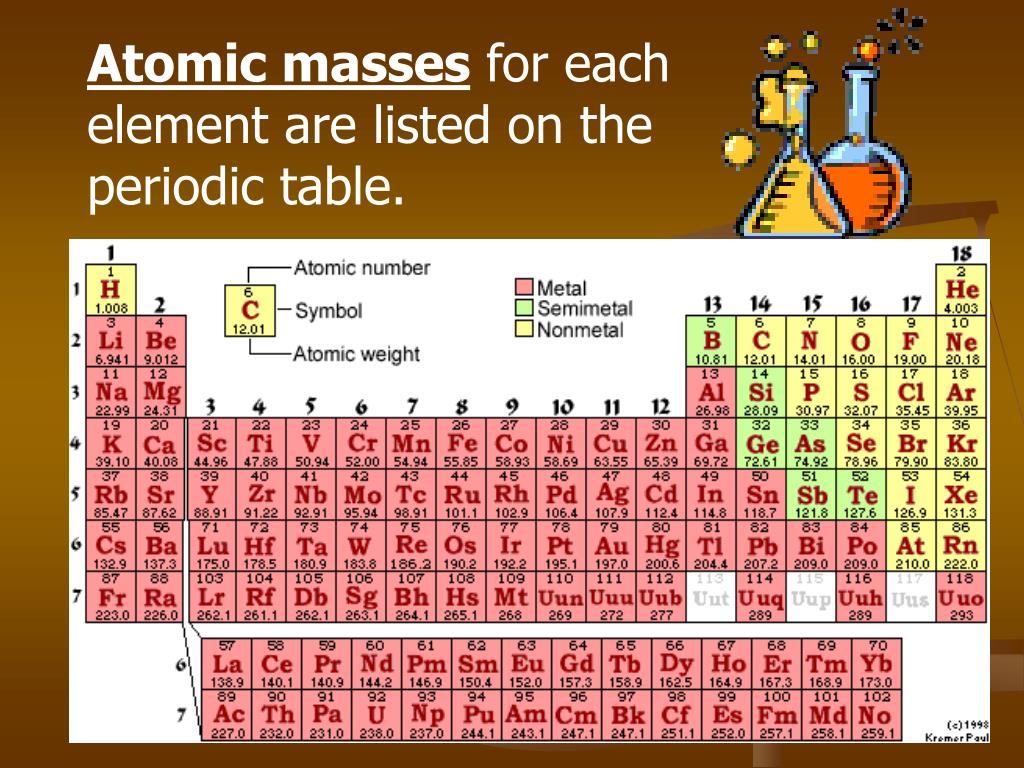
The molar mass of a compound is the mass in grams of 1 mole of that substance, a property expressed in units of grams per mole (g/mol)īecause the definitions of both the mole and the formula mass are based on the same reference substance, 12C, the molar mass of any compound is numerically equivalent to its formula weight in amu. The atomic masses of some elements are known very precisely, to a large number of decimal places. The masses of 1 mole of different compunds, however, are different, since the masses of the individual molecules are drastically different. 207.2 Pb Lead 114 (289) Uuq Ununquadium 10 20.1797 Ne Neon 18 39.948 Ar Argon 36 83.798 Kr Krypton 54 131. The atomic masses in Table A.1 The Basics of the Elements of the Periodic Table represent the number of decimal places recognized by the International Union of Pure and Applied Chemistry, the worldwide body that develops standards for chemistry. Similar to atoms, 1 mole of any compound contains the same number of molecules as 1 mole of any other compund. There are many ways to find the atomic mass of an element, but the easiest way is to look it up on the periodic table of elements. In Chapter 1, we described Dalton’s theory that each chemical compound has a particular combination of atoms and that the ratios of the numbers of atoms of the elements present are usually small whole numbers.\). The Molecular mass of an element can be calculated by adding the atomic masses of each of its constituents. The mole concept can be extended to masses of formula units and molecules as well. 
According to the periodic table, 1 mol of U has a mass of 238.03 g, so the mass of 2 mol is twice that, or 476.06 g. The molar mass of a substance ( M) is the weight of one mole of the substance. Select the correct mass for each of the samples described below. Use the periodic table to determine how many atoms of phosphorus (P) are in a sample that has a mass of 172.90 g. Fe4 Fe (CN)63, NaHCO3, ch3coonh4, h2so4, pb (c2h3o2)23h2o, caso41/2h2o) and press Enter or click Calculate button. Use the periodic table to calculate the molar mass of each of the following compounds. (Since the atomic mass average of sulfur given above only has 3 decimal places, accuracy can not be. The periodic table shows that the atomic mass (rounded to two decimal points) of Al is 26.98, so 1 mol of Al atoms has a mass of 26.98 g. Enter a chemical formula to calculate its molar mass (e.g. The periodic table shows that the atomic mass (rounded to two decimal points) of Al is 26.98, so 1 mol of Al atoms has a mass of 26.98 g. H 2 S 1 O 4 Go to periodic table and determine the atomic mass average (atomic weight) of each element. You can see that The molar mass of Sodium is 22.990 g/mol. So let’s look at the molar mass of Sodium, Sulfur and Oxygen from the above periodic table. Multiply the number of atoms by the atomic weight of each element found in steps 1 and 2 to get the mass of each element in C2H5Cl: Molar Mass (g/mol) C (Carbon) 2 × 12.0107 24.0214. Now in Na2SO4, there are 2 Sodium atoms, 1 Sulfur atom and 4 Oxygen atoms. (8 atoms)(15.9994 amu/atom) = 127.9952 amuįormula mass of Ca 3(PO 4) = 310.177 2amu A tutorial on Molar mass calculations with examples and a molecular weight calculator to aid calculations. You can see the molar mass value of all the atoms from this periodic table. \right ) \right ]=310.177\ amu \notag \) Atoms The molar mass will be equal to: (1 atom x 56 grams/mole Fe) + (2 atoms x 35.5 grams/mole of chlorine) 127 grams/mole of iron (II) chloride. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
 RSS Feed
RSS Feed
